
Trigonal Bipyramidal: Five electrons Examples include PCl5. The variations caused by the lone pair can result in forms like T-shaped, see-saw, and linear.Tetrahedral: Four electron pair Examples include CH4. When there is just one pair, the form changes to trigonal pyramidal (NH3) when it is joined by two pairs of lone pairs, it turns bent or angulated (H2O).Trigonal Planar: Three electron pairs Some examples include BF3. When only one pair occurs, the molecular structure changes and the shape is altered or angular like in SO2.  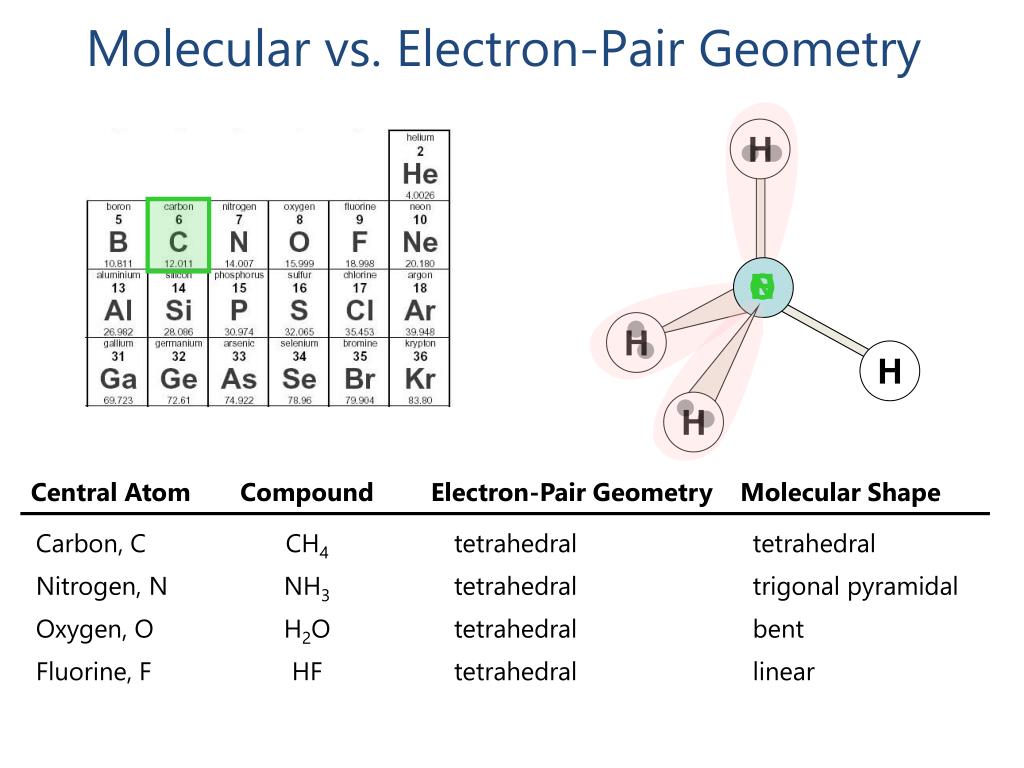
Linear: Two electron pairs Some examples include BeCl2.Common Electron Pair Arrangements & Resulting Molecular Geometries.Lone Pairs vs. Bonding Pairs: Single pairs are more prone to repel as bonding couples. This implies it is possible that the presence of isolated pairs could alter the molecular shape from its electron-based geometrical.Electron Pair arrangement: The spatial arrangement of electron pairs around the center of the atom determines the electron geometry of the molecule.Electron Pair Repulsion: Electron pairs either as bonds (bonding pairs) or on their own (lone pairs) are attracted by each other. They adapt their positions to remain as separated as possible, which reduces Repulsion and thereby stabilizes the molecules.Defined: VSEPR stands for Valence Shell Electron Pair Repulsion Theory. It’s a mathematical model that helps predict the structure of molecules, based on the attraction between electron pairs inside the Valence Shell of the central atom.Applications and Implications of Molecular Geometry and Electron Geometry.Importance in understanding molecular shape and reactivity.Molecular Geometry and Electron Geometry in the comparative chart.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
